EPIDEMIOLOGY
United States poison centers record ~20,000 salicylate exposures annually with ~50–70 deaths. ~4,000 involve children ≤12 years. Pediatric therapeutic exposure and unintentional toddler poisonings have fallen since the 1980s owing to the Reye warning, smaller package sizes, and child-resistant closures. Fatal exploratory ingestions in children are now uncommon.
PHARMACOLOGY
Formulations and sources
Aspirin (acetylsalicylic acid) → rapidly deacetylated to salicylic acid; available as chewable, regular, effervescent, delayed-release, and enteric-coated forms.
Methyl salicylate (oil of wintergreen; 98–100%) is in liniments/ointments and some traditional massage oils. One teaspoon (5 mL) ≈ 7 g salicylate (~22 adult tablets); as little as 4–5 mL can be fatal in a child.
Bismuth subsalicylate (e.g., antidiarrheals): 8.7 mg salicylate per mL; large or chronic use can cause salicylism (and rarely bismuth toxicity).
Magnesium salicylate (OTC analgesic/“water pill” combinations) and topical salicylic acid preparations are additional sources.
Pharmacokinetics/toxicokinetics (high-yield)
Absorption. Therapeutic aspirin peaks in ~1 h for regular tablets; enteric-coated/delayed-release peak later. In overdose, pylorospasm and bezoars delay and prolong absorption; peaks may occur 4–14 h (rarely ≥35 h). Significant percutaneous absorption is uncommon but possible with excessive topicals.
Distribution. Toxicity worsens as salicylate moves from plasma to tissues (e.g., CNS). Protein binding saturates in overdose, increasing the free fraction. Blood pH governs tissue penetration: uncharged HSal crosses membranes; charged Sal− does not. Acidemia increases CNS/renal tubular uptake, amplifying toxicity.
Metabolism. First-order at low concentrations (t½ ~2–4 h); with saturation, zero-order elimination (apparent t½ may approach ~30 h).
Elimination. Renal secretion of salicylate/salicyluric acid dominates; alkaline urine limits back-diffusion and enhances urinary excretion.
TOXIC DOSES (context for exposure history)
Approximate aspirin-equivalent guide: <150 mg/kg none/minimal · 150–300 mg/kg mild–moderate · 301–500 mg/kg severe · >500 mg/kg potentially lethal.
Oil of wintergreen: 5 mL has caused pediatric deaths.
MECHANISMS UNDERLYING CLINICAL EFFECTS
Mitochondrial uncoupling/TCA inhibition → heat (hyperthermia), lactic acidosis, catabolic ketoacids.
Respiratory-center stimulation → tachypnea/hyperpnea and early respiratory alkalosis.
Glycogen depletion/gluconeogenesis impairment → neuroglycopenia (can occur with normal serum glucose).
Gastric irritation/COX inhibition → platelet dysfunction, GI bleeding.
Cochlear effects → tinnitus/hearing changes.
Progressive acidemia raises the HSal fraction → greater CNS/organ penetration; abrupt pH drops (e.g., hypoventilation) can precipitate decompensation.
CLINICAL MANIFESTATIONS
Assessing severity
Use history + exam + acid–base and serial salicylate levels. Clinical status takes precedence over level, especially in cases of chronic exposure/older adults. Early findings can progress despite modest concentrations if acidemia develops or absorption is delayed.

Pattern highlights
Tachypnea/hyperpnea with large tidal volumes is the hallmark and early sign.
Hyperthermia is later and correlates with severity; absence does not exclude toxicity.
Tinnitus/hearing changes can occur at 15–30 mg/dL.
GI: pylorospasm, delayed emptying, vomiting; GI bleeding in severe cases.
Fluids/electrolytes: large insensible/urinary losses; whole-body K⁺ depletion despite normal serum value; hypo- or hypernatremia may occur.
Acid–base: early respiratory alkalosis → mixed disorder → acidemia if severe.
CNS: agitation → confusion → seizures/coma; CNS toxicity defines severe poisoning and may occur despite falling/near-therapeutic serum levels (tissue burden).
Pulmonary edema (noncardiogenic) is severe—more frequent in older adults/chronic exposure.
Other: transient hyperglycemia- then hypoglycemia; leukocytosis; platelet dysfunction/coagulopathy; rare dysrhythmias; distributive physiology; reversible hepatic injury.
DIFFERENTIAL DIAGNOSIS (what salicylism can mimic)
DKA (consider euglycemic DKA with SGLT2 inhibitors)
Sepsis/pneumonia/SIRS (salicylate pulmonary edema can mimic infection)
Toxic alcohols (methanol/ethylene glycol)
Acute iron poisoning
Ethanol (children)
Key tip: In unexplained metabolic acidosis, measure a serum salicylate—even if the anion gap is normal (pseudohyperchloremia can artifactually lower the gap on some analyzers).
EVALUATION AND DIAGNOSTIC TESTING
History & exam (focused)
Dose/formulation and timing; intent; co-ingestants; chronic aspirin/OTC salicylate use. Symptoms: tinnitus, hearing change, vomiting, dyspnea, agitation/confusion. Examine respiratory pattern (rate/depth), oxygenation, temperature, volume status, and mental status; reassess frequently.
Laboratory studies (initial set)
Serum salicylate; electrolytes (Na, K, Cl, HCO3), Mg, phosphate, glucose; BUN/Cr; ABG/VBG; serum lactate; urinalysis with urine pH; acetaminophen level; CBC; PT/INR/PTT; liver enzymes; pregnancy test when applicable; ECG; CXR if hypoxemia/rales; head CT if unexplained AMS/focal findings.
Serum salicylate concentration—how to interpret
Toxic concentration: most symptomatic at >30–50 mg/dL (2.2–3.6).
Serial testing: repeat every ~2 h initially (every 1–2 h if clinically worsening) to capture delayed/prolonged absorption and evolving tissue distribution; levels lag behind clinical status.
Unit pitfalls: some labs report mg/L (tenfold higher than mg/dL). Lipemia can spuriously elevate spectrophotometric assays; diflunisal may interfere.
Done nomogram: obsolete with modern formulations/kinetics.
DIAGNOSIS
Suspect with compatible history or the classic triad of tachypnea/hyperpnea, tinnitus, and acid–base disturbance (early respiratory alkalosis, later mixed/metabolic acidosis). Confirm with >30 mg/dL, recognizing poor correlation between a single level and severity—particularly in chronic toxicity. Determine acute vs chronic; diagnostic delay increases mortality.
SPECIAL POPULATIONS
Children may be harder to recognize (tachypnea/irritability common). Chronic pediatric toxicity tends to be more serious at a given level; mixed or pure metabolic acidosis and loss of respiratory drive are more likely than in adults. Noncardiogenic pulmonary edema is less common in children but ominous when present.
OVERVIEW OF MANAGEMENT
See: Comprehensive Guidelines for Managing Poisoning in Adults
Salicylate poisoning: Rapid overview of emergency management
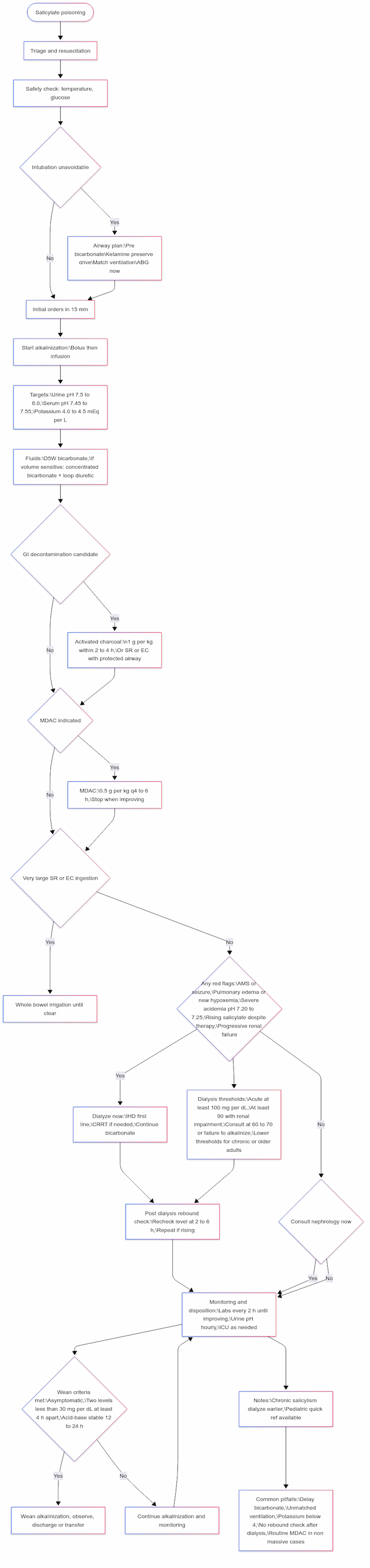
Patients with severe salicylate toxicity can deteriorate rapidly and be challenging to manage. Principles mirrored in the flowchart:
ABCs first. Monitor continuously, place two large-bore IVs, maintain NPO status, and administer an antiemetic.
Airway caution. Avoid tracheal intubation if possible. If unavoidable (e.g., seizures, procedures, ventilatory failure): pre-bicarbonate, prefer agents that preserve ventilatory drive (e.g., ketamine), match or exceed pre-tube minute ventilation, and obtain an ABG immediately post-tube. Anticipate early dialysis in severe cases.
Initial orders (≤15 min). Salicylate level, BMP (with K⁺), glucose, creatinine, lactate, ABG/VBG, acetaminophen level, EKG; CXR if respiratory symptoms; pregnancy test when relevant. Repeat key labs every 2 hours until clearly improving.
Start alkalinization early. NaHCO₃ 1–2 mEq/kg IV bolus, then 150 mEq NaHCO₃ in 1 L D5W at ~200–300 mL/h. Targets: urine pH > 7.5–8.0; serum pH ~ 7.45–7.55. Maintain K⁺ 4.0–4.5 mEq/L.
See: Antidote
GI decontamination (selective). Single-dose AC 1 g/kg (max 50 g) if ≤ 2–4 h from ingestion or SR/EC formulation with protected airway. MDAC is only for massive or SR/EC ingestions or rising levels despite therapy. WBI is only for very large SR/EC ingestions with a protected airway.
Identify red flags (any time). Altered mental status/seizures, pulmonary edema or new hypoxemia, severe acidemia (~ pH ≤ 7.20–7.25), rising level despite therapy, progressive renal failure, or fluid overload precluding alkalinization.
Dialysis decisions. Use the Dialysis Decision Support (EXTRIP-based) table below for immediate indications, level-based thresholds (supportive only), early nephrology consult triggers, and modality/rebound guidance. When the criteria are met, proceed directly to IHD (CRRT only if IHD is unavailable/contraindicated) while continuing serum/urine alkalinization during the arrangements.

Post-dialysis rebound. Re-check salicylate 2–6 h after IHD; repeat/extend dialysis if rising or symptomatic.
Wean criteria. Asymptomatic and two levels < 30 mg/dL ≥ 4 h apart, with acid–base stable 12–24 h (longer if chronic).
Consult early. Engage with medical toxicologist/poison centers, as well as nephrology, at the outset.
ASSESS SEVERITY OF TOXICITY
Assess severity using signs/symptoms, serum salicylate trajectory, and acid–base status; do not rely on a single serum concentration (levels commonly lag clinical status). Toxicity evolves over time: early respiratory alkalosis may progress to a mixed disorder and then acidemia as absorption continues or tissue distribution increases. Factors such as enteric-coated formulations, bezoars, pylorospasm, and co-ingestants can delay onset and prolong absorption.
General staging framework (clinical effects):
Mild (early). Tinnitus, nausea, and tachypnea may be present. Often corresponds to 30–50 mg/dL (2.2–3.6 mmol/L); blood pH normal or alkalemic; urine pH variable early.
Moderate (middle). Mild hyperthermia, tachycardia, hyperpnea, vomiting, diaphoresis; mental status preserved; typically 50–90 mg/dL (3.6–7.2) (can be lower in chronic exposure). Blood pH is alkalemic or mixed.
Severe (late). Any CNS change, pulmonary or cerebral edema, severe acidemia, or other dialysis indications. Often ≥ 90–100 mg/dL in acute cases (can decompensate at lower levels); blood pH acidemic (< 7.30) or mixed.
MANAGEMENT IN MOST PATIENTS
ABCs and supportive care
Airway. Avoid intubation if possible. If needed: NaHCO₃ 1–2 mEq/kg IV bolus before induction; consider awake/ketamine-facilitated approach to preserve drive; minimize apnea; after placement, match/exceed pre-tube minute ventilation and re-check ABG at 15–30 min.
Oxygenation. Supplemental O₂ as needed; pulmonary edema may require escalating support.
Blood pressure support. Resuscitate with crystalloid as needed; use vasopressors (e.g., norepinephrine/phenylephrine) if hypotension persists due to distributive physiology.
Gastrointestinal decontamination
Single-dose AC as above. Do not intubate solely to give AC.
MDAC (selective). Only for massive/SR-EC ingestions or rising levels despite optimal therapy; 0.5 g/kg q4–6 h (max 50 g/dose). Hold for ileus, GI bleeding, or aspiration risk; stop once the condition improves.
WBI. Rare; consider only for very large SR/EC ingestions with a protected airway after discussion with toxicology.
Frequent laboratory monitoring
q2 h: salicylate, electrolytes (with K⁺), and blood gas until clearly improving.
Hourly: urine pH while alkalinizing.
Glucose hourly if altered mental status or hypoglycemia; treat neuroglycopenia even with “normal” serum glucose.
Fluid and electrolyte repletion
Resuscitate to euvolemia; several liters may be required in severe cases (individualize if pulmonary/cerebral edema risk).
Potassium: maintain K⁺ 4.0–4.5 mEq/L (aggressively replete if < 4.0 or falling during alkalinization). Replete Mg/PO₄ as needed, especially with ongoing bicarbonate.
Serum and urine alkalinization
Bolus: NaHCO₃ 1–2 mEq/kg IV.
Infusion: 150 mEq NaHCO₃ in 1 L D5W at ~200–300 mL/h; titrate to urine pH > 7.5–8.0 and serum pH ~ 7.45–7.55.
Add 20–40 mEq/L KCl (or K-phosphate) to maintain the K⁺ target.
If serum pH > 7.55–7.6 or hypernatremia develops, reduce rate/concentration (e.g., 50–100 mEq/L in D5W).
INDICATIONS FOR HEMODIALYSIS
Dialyze now (irrespective of level) for:
Altered mental status (delirium, agitation, lethargy, seizure)
Severe acidemia (~ pH ≤ 7.20–7.25, or < 7.30 and worsening despite therapy)
Pulmonary edema / new hypoxemia; cerebral edema
Rising salicylate despite optimized alkalinization
Progressive renal failure/clinical deterioration (shock, dysrhythmia)
Fluid overload precluding alkalinization
Consult nephrology now at ≥ 60–70 mg/dL, suspected chronic toxicity, AKI, failure to achieve/maintain alkalinization, or significant symptoms.
Level thresholds (supportive, not standalone):
Acute, normal renal function: ≥ 100 mg/dL
Acute with renal impairment: ≥ 90 mg/dL
Chronic / older adults: ≥ 60 mg/dL with symptoms (use a lower threshold)
Modality & follow-up
IHD is the first-line treatment; CRRT is only used if IHD is unavailable/contraindicated (due to slower clearance).
Post-dialysis rebound: Re-check salicylate levels at 2–6 hours; repeat/extend dialysis if levels are rising or symptoms persist.
Bicarbonate during IHD: pause or adjust to avoid over-alkalemia; restart as needed until stable.
See: Hemodialysis in Poisoning: A Comprehensive Guide for Healthcare Providers
MILD TOXICITY
Observation with close q2 h labs is acceptable if asymptomatic/mild and trending down. Give single-dose AC if a plausible GI burden remains (≤ 2–4 h window or SR/EC + protected airway). Escalate immediately if any red flag appears.
MODERATE TOXICITY
Definition (context): Persistent/worsening symptoms (e.g., hyperpnea, diaphoresis, vomiting, mild hyperthermia; MS preserved). Often corresponds to ~50–90 mg/dL (3.6–7.2 mmol/L) in acute exposure but can occur at lower levels in chronic exposure.
Management (per Figure 1):
Site of care: ED with low threshold for ICU if trajectory uncertain or red flags appear.
Monitoring cadence: q2 h salicylate/electrolytes/gases; hourly urine pH while alkalinizing; hourly glucose if MS changes or hypoglycemia.
Alkalinization early: NaHCO₃ 1–2 mEq/kg IV bolus, then 150 mEq NaHCO₃ in 1 L D5W @ ~200–300 mL/h.
Targets: urine pH >7.5–8.0; serum pH ~7.45–7.55.Potassium goal: K⁺ 4.0–4.5 mEq/L (aggressively replete if <4.0 or falling); replete Mg/PO₄ as needed.
GI decontamination (selective):
AC single dose (1 g/kg, max 50 g) if ≤2–4 h or SR/EC + protected airway.
MDAC only if massive or SR/EC ingestion or rising levels despite therapy (0.5 g/kg q4–6 h, max 50 g/dose). Hold for ileus/aspiration risk; stop once improving.
Nephrology consult early if any of: level ≥60–70 mg/dL, suspected chronic exposure, AKI, failure to achieve/maintain alkalinization, or significant symptoms.
Escalation trigger: Any red flag (AMS/seizure, pulmonary edema/new hypoxemia, severe acidemia, rising level despite therapy, progressive renal failure) → treat as Severe.
SEVERE TOXICITY (ACUTE)
Definition: Any CNS change, pulmonary edema/new hypoxemia, severe acidemia (~pH ≤7.20–7.25 or <7.30 and worsening), rising level despite therapy, renal failure/clinical deterioration—regardless of dose. Often ≥90–100 mg/dL in acute cases, but decompensation can occur at lower levels.
Immediate actions (per Figure 1):
ICU level care; continuous monitoring.
Airway caution: Avoid intubation if possible. If unavoidable: pre-bicarbonate, prefer ketamine (preserve drive), minimize apnea, match/exceed pre-tube minute ventilation, ABG at 15–30 min.
Continue/optimize alkalinization: As above; ensure K⁺ 4.0–4.5 mEq/L; correct Mg/PO₄.
Dialyze now for any severe criterion above, or acute level ≥100 mg/dL (≥90 mg/dL with renal impairment).
Modality: IHD first-line (CRRT only if IHD unavailable/contraindicated).
Rebound: Re-check salicylate at 2–6 h post-IHD; repeat/extend if rising or symptomatic.
Bicarbonate during IHD: pause or adjust to avoid over-alkalemia; restart after as needed until stable.
Oxygenation/ventilation: Supplemental O₂; manage pulmonary edema; avoid hypoventilation (pH drop → tissue HSal surge).
Ongoing care:
q2 h salicylate/electrolytes/gases; hourly urine pH while alkalinizing; hourly glucose if AMS/hypoglycemia.
Wean criteria (for de-escalation/discharge planning): Asymptomatic and two levels <30 mg/dL ≥4 h apart, with acid–base stable 12–24 h (longer if chronic).
CHRONIC SALICYLISM (INCLUDING ACUTE-ON-CHRONIC)
Definition : >100 mg/kg/day for ≥2 days or repeated dosing (often older adults). Clinical severity may be disproportionate to level (e.g., significant toxicity at 30–60 mg/dL).
Management nuances (per Figure 1):
Lower threshold to escalate/dialyze: Favor earlier dialysis—e.g., ≥60 mg/dL with symptoms, or any severe criterion (AMS, pulmonary edema/new hypoxemia, severe acidemia, rising level despite therapy, renal failure).
Same alkalinization/K⁺ strategy as acute; don’t “chase” urine pH indefinitely if patient is deteriorating—dialyze sooner.
Monitoring cadence: As above (q2 h levels/gases/electrolytes; hourly urine pH while alkalinizing).
Disposition: Expect longer stabilization window before wean criteria (asymptomatic, two levels <30 mg/dL ≥4 h apart, stable 12–24 h or longer).
INEFFECTIVE THERAPIES / AVOID
Forced diuresis or routine diuretics—do not improve clearance beyond alkalinization and increase edema risk.
Acetazolamide—raises urine pH but lowers blood pH (promotes tissue penetration of salicylic acid).
DISPOSITION
Per the flowchart wean criteria, consider discharge/transition when the patient is asymptomatic, vital signs and exam are normal, two salicylate levels are < 30 mg/dL at least 4 hours apart, and acid–base is stable for 12–24 hours (longer if chronic exposure). Otherwise, manage in an ICU for frequent reassessment and potential dialysis.
ADDITIONAL RESOURCES
Regional poison centers (US): 1-800-222-1222 (24/7). Many hospitals have bedside medical toxicology consult services.
Toxicology and nephrology consultation early in the course is strongly recommended.
